
So, if the atomic weight is 201, we subtract 80 and get the number 121. Remember that the atomic weight minus the atomic number (number of protons) gives us the number of neutrons in an atom. Therefore, if it has 80 electrons, it must have 80 protons. If the Mercury (Hg) atom is neutral, it has the same number of electrons as protons. Look up its atomic number (47, which is the number of protons it has) and then, if there is no charge on the atom, you know you have the same number of electrons as protons. Therefore the atomic weight is derived from the average number of neutrons that present in a type of atom.Ĥ. Atomic number (Z) is the number of protons contained in the nucleus of a certain element. So scientists have determined the average number of neutrons in an element and this gives us an average number with decimal points. Because each element has the exact same number of protons in all its atoms but different number of neutrons. This final number tells you how many neutrons you have.ģ. if you subtract the atomic number from the atomic weight, the number you get is rounded (up or down) to the nearest number. the atomic number is the one on top of the letter and it gives us the number of protons in an atom.Ģ. Determine how many protons and neutrons it has, then write it out using the subscript/superscript format.ġ. A neutral atom of Mercury (Hg) has an atomic weight of 201 and contains 80 electrons. 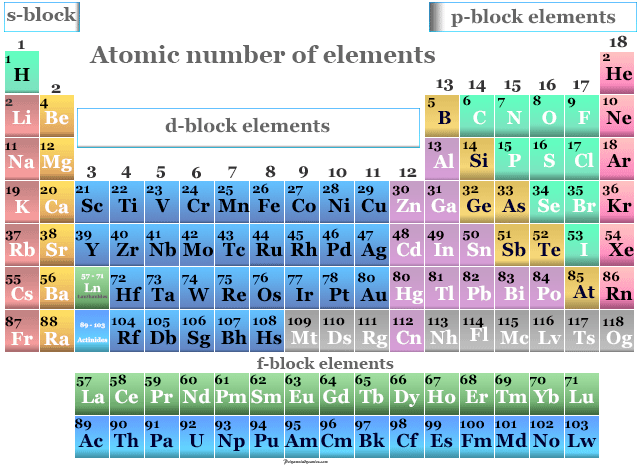
How can you tell how many electrons the element of silver (Ag) has?ĥ. Why do atomic weights have decimal points?Ĥ. How can I tell how many neutrons an atom has by looking at the periodic table?ģ. Which is the atomic number and what does it tell us?Ģ.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
